Precision Medicine and Its Emerging Role in Clinical Trials
“Precision medicine (generally considered analogous to personalized medicine or individualized medicine) is an innovative approach that uses information about an individual’s genomic, environmental and lifestyle information to guide decisions related to their medical management. The goal of precision medicine is to provide a more precise approach for the prevention, diagnosis and treatment of disease.” (National Human Genome Research Institute)
At its core, pharmacogenomics (PGx) aims to maximize therapeutic efficacy while minimizing adverse effects. Historically, clinical trials have been designed to determine what works for a representative patient population. However, we now understand that genetic variability can significantly influence how patients respond to the same therapy.
Precision medicine has been called “the future of healthcare” for over two decades. Today, it is finally becoming a reality and shaping how clinical trials are designed, executed, and interpreted.
From Discovery to Reality
The concept of variability in drug response is not new. Enzyme polymorphisms such as G6PD deficiency were identified in the 1950s, followed by the discovery of CYP2D6 polymorphisms in the 1970s, both demonstrating meaningful differences in how patients metabolize medications.
The true inflection point came with the launch of the Human Genome Project in 1990, completed in 2003. This milestone generated widespread excitement, positioning personalized medicine as the future of healthcare.
So why, more than two decades later, is precision medicine still not fully mainstream?
The answer may lie in scalability.
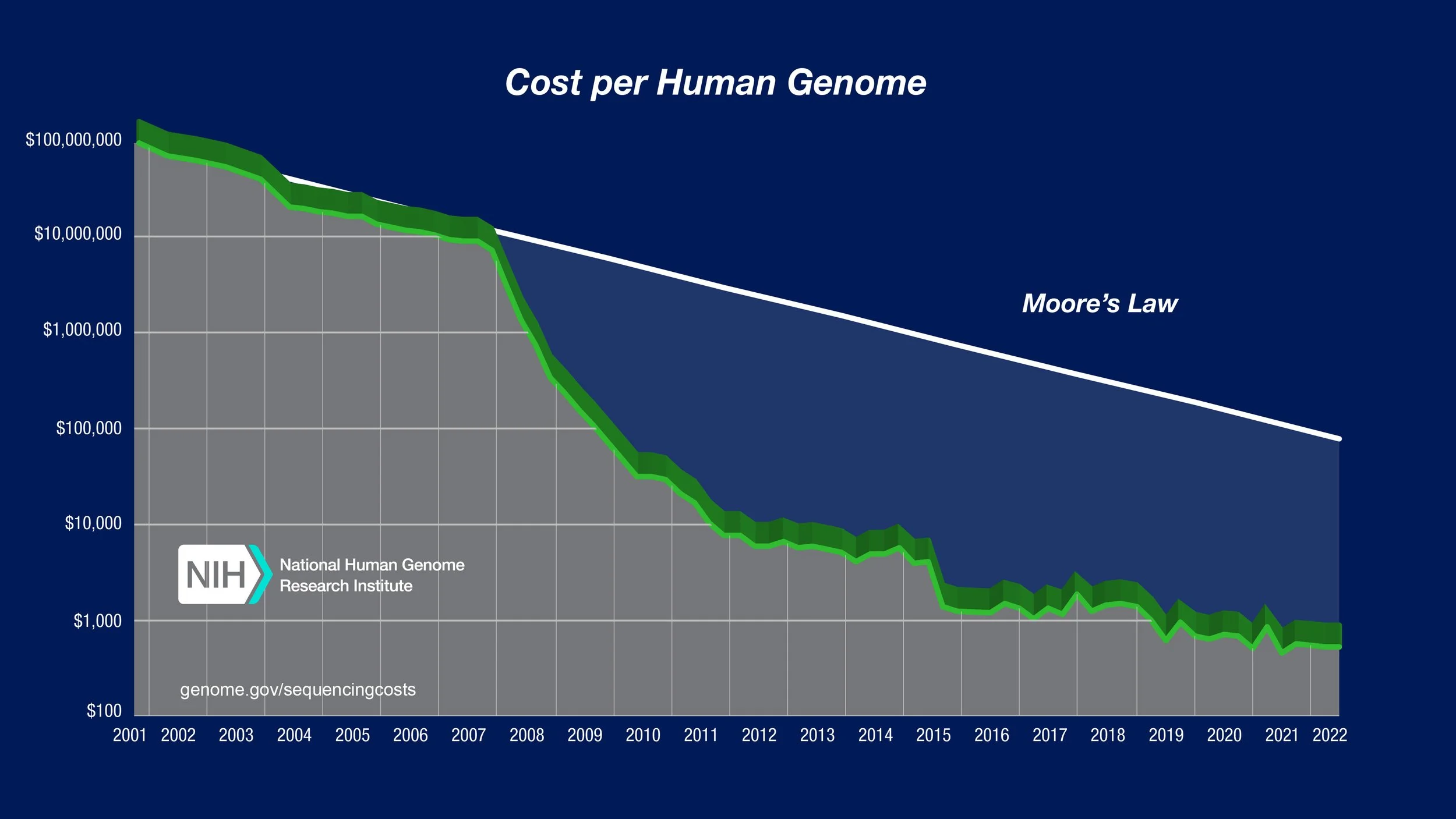
While the Human Genome Project provided the foundational reference needed to enable pharmacogenomic testing, the cost of sequencing remained a major barrier. Sequencing during the project cost approximately $2.7 billion. Today, that cost has dropped dramatically to approximately $200 to $1,000 per genome, representing a reduction of over 99.99%.
Source: genome.gov/sequencingcosts
This shift has transformed precision medicine from a theoretical concept into a practical tool. In parallel, the cost of pharmacogenomic testing has decreased significantly, and reimbursement is expanding, with some states even mandating insurance coverage for specific use cases.
In recent years, several factors have accelerated the adoption of precision medicine. Advances in next-generation sequencing, increased availability of real-world data, and growing regulatory support for biomarker-driven development have all contributed to making precision approaches more feasible at scale. What was once cost-prohibitive and operationally complex is now becoming increasingly embedded in standard development strategies.
Precision Medicine in Today’s Clinical Trials
Precision medicine is no longer limited to theory. It is actively shaping modern drug development.
Many of today’s most impactful therapies are rooted in targeted approaches. In oncology, for example, treatments such as immune checkpoint inhibitors are used in biomarker-defined patient populations, improving outcomes by aligning therapies with underlying disease biology.
A well-known example of precision medicine in practice is HER2-positive breast cancer, where patients are selected for targeted therapy with trastuzumab based on HER2 overexpression. Similarly, EGFR mutations in non-small cell lung cancer guide the use of EGFR inhibitors, significantly improving response rates compared to non-targeted approaches.
These examples highlight how biomarker-driven strategies can transform both clinical outcomes and trial design by ensuring therapies are matched to the patients most likely to benefit.
However, precision medicine is not only about identifying who should receive a therapy. It is also about understanding who should not, and why.
This is where pharmacogenomics plays an increasingly important role in clinical trials.
Incorporating PGx into trial design, even at an exploratory level, can:
Identify subpopulations with enhanced or diminished response
Explain variability in efficacy and safety outcomes
Inform dose optimization strategies
Reduce late-stage trial failures by improving patient selection
This shift is also changing how trials are structured. Enrichment strategies, adaptive trial designs, and basket or umbrella trials are becoming more common as sponsors seek to evaluate therapies within genetically defined populations. These approaches can improve trial efficiency, but they also require more sophisticated planning, data integration, and operational execution.
Rather than treating variability as noise, precision medicine allows sponsors to treat it as actionable data.
Operational Implications for Sponsors and CROs
As precision medicine becomes more integrated into clinical development, it introduces new layers of complexity:
Smaller, more targeted patient populations requiring refined recruitment strategies
Biomarker-driven endpoints increasing reliance on specialized assays and diagnostics
Greater data integration needs combining genomic, clinical, and real-world data sources
For sponsors and CROs, success will depend not only on scientific innovation, but on operational execution.
Clinical trials must now be designed with precision in mind from the outset. This includes incorporating biomarker strategies, ensuring appropriate infrastructure for genomic data handling, and aligning with evolving regulatory expectations.
Looking Ahead
Precision medicine is no longer “the next big thing.” It is actively reshaping how therapies are developed and delivered.
As sequencing becomes more accessible and data capabilities continue to expand, the role of precision medicine in clinical trials will only grow.
The organizations that succeed will not be those that simply adopt precision medicine, but those that can operationalize it effectively. Translating complex biomarker strategies into executable, compliant, and scalable trials will be a key differentiator in the next generation of drug development.